Welcome to this page on LaNts! I’ve written this as I appreciate that many/most/all of you won’t have heard about LaNts unless you have spoken to myself or one of my team in the last few years! This page aims to give you some context and hopefully help you appreciate why we are so interested in this little known but pretty awesome protein family.
NB For some of this to make sense it might help to know a little bit about laminins. However, we have that covered; this link will take you to our primer on laminins or you can watch this video.
LaNt = Laminin N terminal proteins
The LaNts are a family of smallish proteins (50-100kDa) that get made from the same genes as laminins through a process called alternative splicing (technically intron retention and alternative polyadenylation – our animation about this on youtube here). What this means is that they although they share features with the laminins, they miss out on some of the key features that make laminins laminins.
Why have you not heard about LaNts?
Well, actually very little is known about these proteins. Currently there is only one paper published about them (available here) and in that paper we did little more than describe the existence of this protein family.
The discovery story isn’t published anywhere (I might blog about this one day). It is somewhat serendipitous. We were trying to find the cause for a rare genetic skin disorder during my PhD, and for a bunch of reasons that won’t become obvious just now, we were looking really closely at the LAMA3 gene. At that time the human genome project wasn’t as complete as it is now and so things weren’t fully annotated. It was whilst we were deep in the data trying to work out what was real and what was an artefact that we noticed the presence of a group of mRNAs that looked like the real deal. Importantly, my supervisor, Prof Irwin Mclean, was prepared to let me break from my main PhD plan to follow-up this finding and take the research forward to where it is now. Irwin was a fantastic boss, he let me develop my scientific thinking and follow the data wherever it would take me; even allowing me to look at the genetic history of the disease we were studying (read about how Mongol invasion’s were relevant here!).
After finishing my PhD, I moved to Northwestern University, Chicago to work with the world-renowned laminin expert- Jonathan Jones. Although I was able to get my PhD work published at this time, I mostly worked on hemidesmosomes and laminins (published work here). With Jonathan’s help I secured some funding to continue with the LaNt work in the last year at NU and subsequently established myself as an independent faculty member at the University of Liverpool. International moves and the challenges of getting funding have meant there was a lag in progress but I now have had a small team working on the LaNts for a few years and the story is beginning to move forward.
In the “Hamill lab” we are trying to expand what is understood about this “new” family of proteins, studying everything* about them. Right now we have people investigating them at many levels; from their distribution in the body, their function at the protein, cell, tissue and organism level, all the way through to investigating how the unusual splicing events that leads to their generation are regulated and whether we can target aspects of their biology in therapies (read about our cancer project here).
Fantastic proteins and where to find them
We predict that the role of the different LaNt proteins depends on what else is going in the tissue at the time. I’ll go a bit deeper into this shortly but, for now, our data indicate that LaNts provide a way of changing cellular responses to laminins and this in turn means that they could influence any of the diverse array of different functions that have been attributed to laminins. These functions include controlling wound repair, cancer invasion and metastasis, blood vessel growth, stem cell differentiation and lots of the tissue remodelling that goes on during embryonic development, in the hair cycle and mammary gland maturation. This list goes on; laminins do a lot of cool stuff!
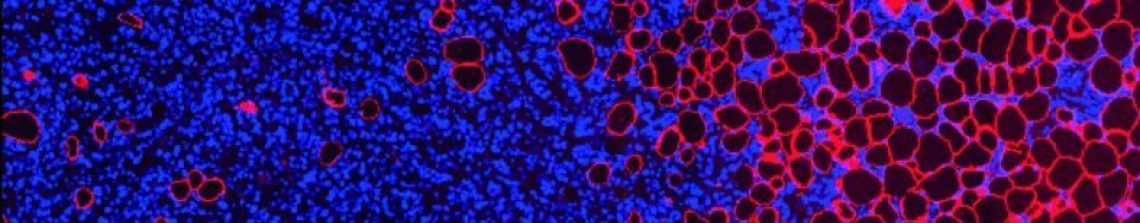
From these comments, comes the question; where do you find LaNts? We’re working toward answering this question just now. Over the last few years we have raised antibodies against human and mouse forms of one LaNt protein, LaNt α31. With these tools in place we are asking where and when is this protein expressed. The first manuscript from this work is out for review at the moment and I will update this page again once it is published. For now here’s a pretty cool panel from one of the figs…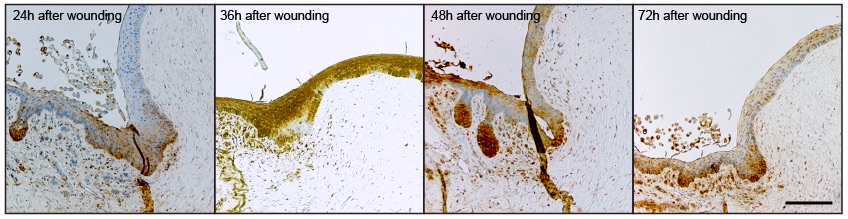 In the pic above we are looking at part of the front of the eye that we call the limbus. The brown colour is where our anti-LaNt antibodies have bound to the tissue. The blue dots (you’ll have to zoom in to see them as dots!) are each nuclei. What we see are groups of cells that are really brown, i.e. have lots of LaNt, whereas other cells don’t express the protein.
In the pic above we are looking at part of the front of the eye that we call the limbus. The brown colour is where our anti-LaNt antibodies have bound to the tissue. The blue dots (you’ll have to zoom in to see them as dots!) are each nuclei. What we see are groups of cells that are really brown, i.e. have lots of LaNt, whereas other cells don’t express the protein.
What does these findings mean?
Good question, but the limbus is an interesting place! It’s the boundary between the conjunctiva (which makes the tear film) and the cornea which is the transparent front part through which light passes. The limbus main jobs are to keep the conjunctiva from growing over the cornea, and to replace the cells in the cornea (the limbus contains stem cells). The pics above are various stages after a wound has been introduced so we are looking at the limbus at a time when the stem cells have been activated in order to replace the damaged cornea. The strong LaNt staining could be an indicator that this protein is involved in the repair process.

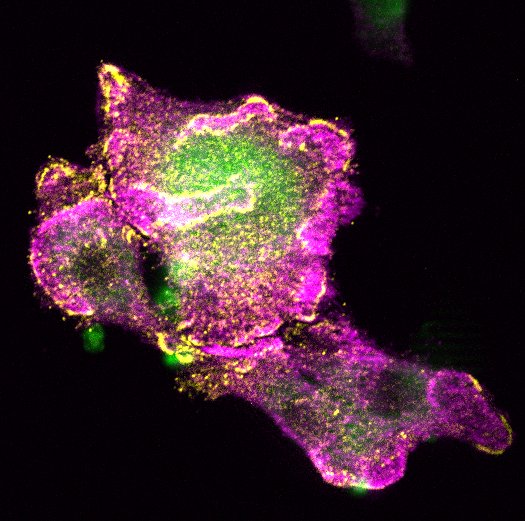
Another area where we have made interesting observations is in cancers arising from epithelial tissues (squamous cell carcinomas). The picture to the above is from a patient with a squamous cell carcinoma on their tongue. What we have seen is a big increase in LaNt staining in the cancerous epithelium compared with the normal epithelium. We don’t yet know exactly what this means for patients but our current work is aimed at finding that out, finding out what controls the expression and whether we can turn the LaNt expression off again. The first manuscript describing this is being prepared at the moment so hopefully you can read about it in the primary literature soon but for now here is our lay-friendly page on our splicing switch and cancer project.
What do LaNts do?
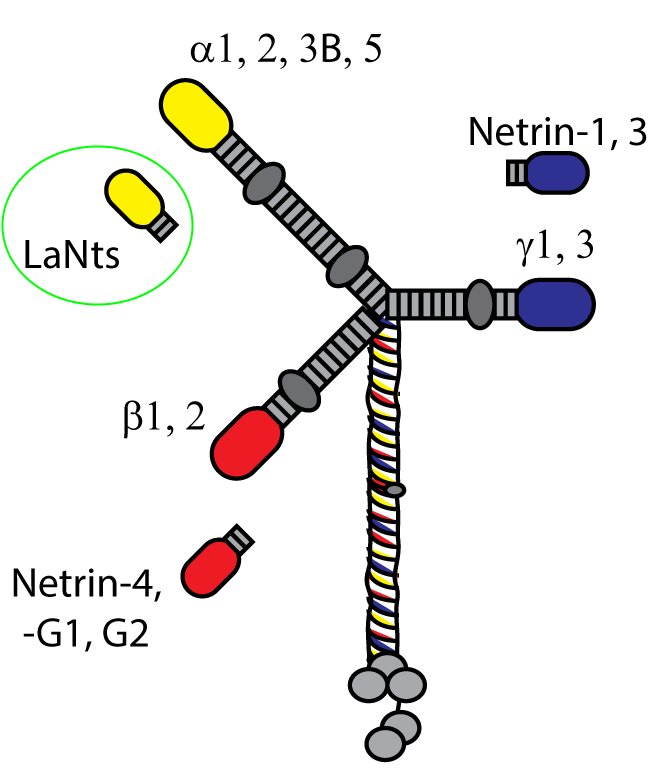
LaNt stands for “laminin N terminus” which reflects the major structural element of the proteins. Each LaNt consists of a signal sequence followed by a globular domain termed a laminin N terminal domain (LN domain, previously known as domain VI), a short stretch of laminin-type epidermal growth factor-like repeats (LE domain) and carboxyl terminal domain of no known function. All in all this gives proteins of 50-100kDa, about one tenth of the size of an assembled laminin. This size and structure is quite similar to that of the netrins (see diagram above) which make us think that some things the netrins do could also be done by the LaNts.
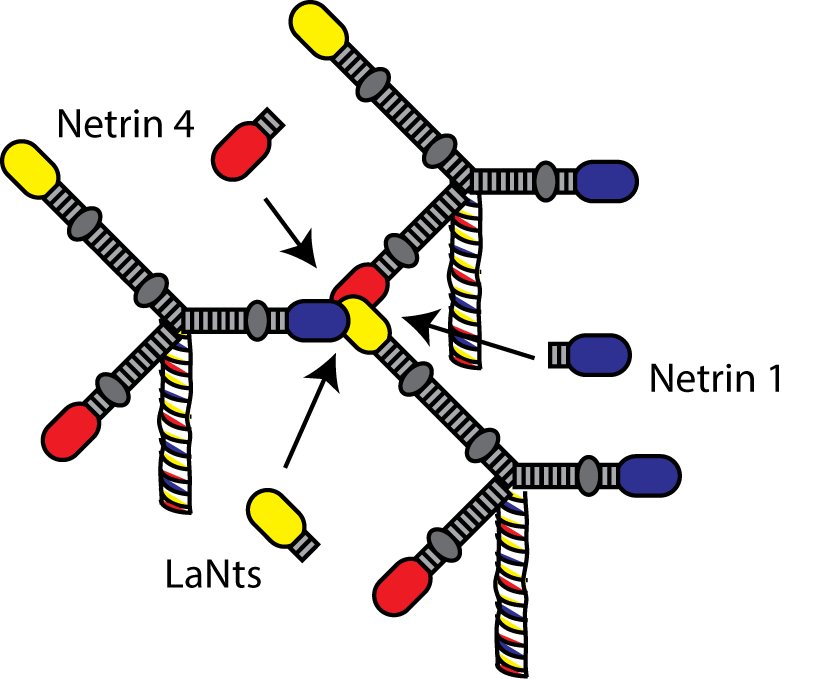
Of the domains found in LaNts, our current thinking is that it is the LN domain that is likely the most important in terms of protein function.
LN domains have been studied quite a lot in the laminins. Some really elegant studies by Peter Yurchenco and Erhard Hohenester have shown that LN domains are the regions through which interaction between laminins occur, which ultimately allow the formation of laminin polymers or networks. Further studies have revealed that formation of laminin polymers requires interaction between three LN domains. As the LaNt proteins contain a LN domain too we think that the LaNts may compete with laminins at these LN to LN interaction sites, either preventing network assembly in the first place or disrupting already formed networks.
There is a precedent for this idea. Work in the netrin field has shown that this sort of inhibition of network assembly occurs with netrin-4 and this has implications for blood vessel and nerve growth (2016 Nature Comms paper by Raphael Leuven et al)
Path or stepping stones?
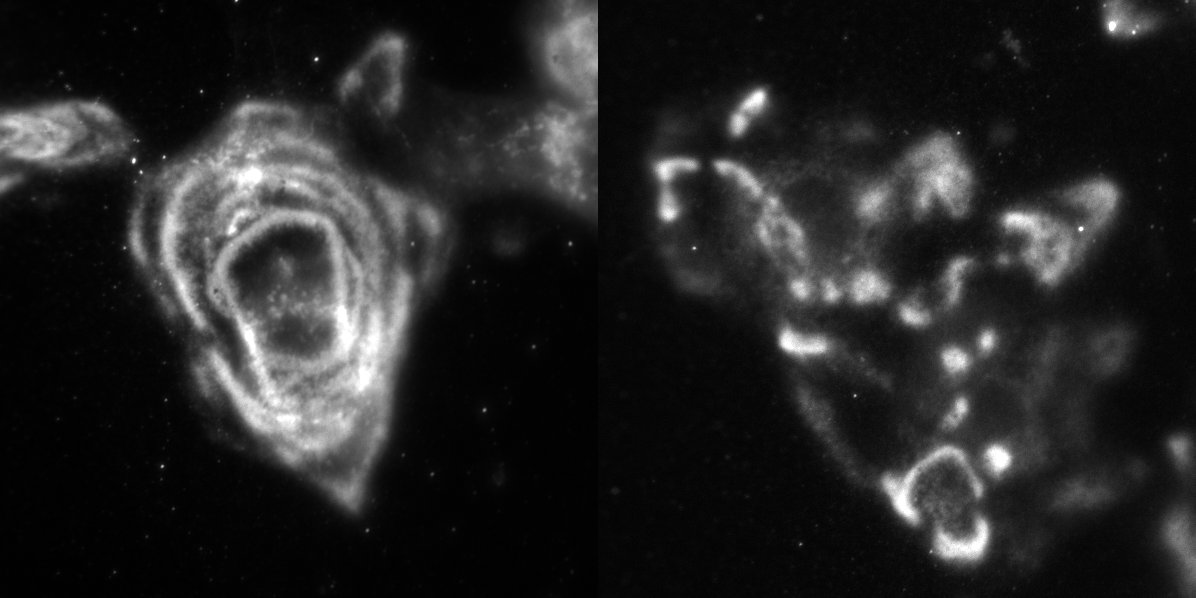
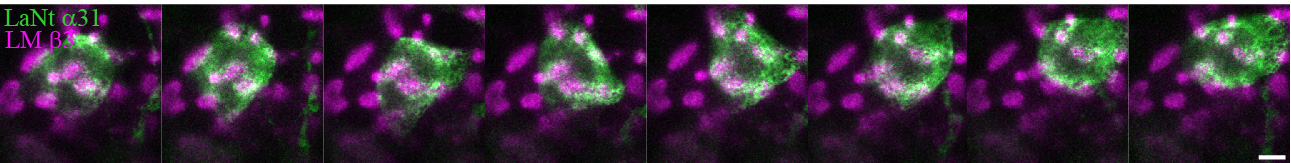
Consistent with our hypothesis, data from our lab has revealed that when we engineer skin or corneal cells to have more LaNt than usual, the laminin is assembled in an aberrant way compared with control cells (if normal laminin is assembled into swirls or path-like patterns then the LaNt overexpressing cells assemble the same protein into more stepping stone-like pattern). You can see examples of this in the picture.
You can also see this in action as time-lapse video below; with LaNt (on the left) and laminin (right) getting deposited by a corneal epithelial cell. In this experiment we are watching the cells not long after we have plated them onto glass dishes and you can see how the two patterns that are left behind as the cell moves are very similar – we currently think they come out at the same time in the same place – and other data that we have indicates that the two proteins are capable of interacting directly.
Importantly all these effect mean that changing the levels of LaNts up or down could provide a control mechanism to change cell behaviour – you can see this in action in the movie below where we have taken corneal epithelial cells and introduced a scratch, then filmed them as they close the “wound”. The cells on the right have too much LaNt and you can see them struggle to get anywhere… their migration is impaired. Changing LaNt levels could mean that stationary cells can start moving or cause moving cells to stop (eg once a wound is healed).
These findings are part of a third manuscript that we are completing at the moment, I will update this page once that paper is out so you can see the whole data set. In case you wondered, yes it seems like a lot of our papers are going to come out at the same time! you can sign up for alerts below and we’ll keep you posted when the papers hit the presses.
Goldilocks?
While what I have described so far seems nice and simple in principle, we actually also have data that shows that too little LaNt can be a problem as well as too much. This suggests that it isn’t merely a yes/no binary-type interaction but more of a “Goldilocks” type affair where a cell needs just the right amount of LaNt to control its behaviour. I did mention these proteins are super cool! This is leads to questions about how the production rates of these proteins are controlled so, you’ve guessed it, we are working on that too! It really is a fun time in the Hamill lab!
To bring this back to the real world again, we already know that laminins are super important in terms of controlling lots of aspects of cell behaviour and our work is showing that LaNts change laminins and that LaNt abundance changes in different tissue contexts so it is looking increasingly likely that LaNts are important regulators of a bunch of different things. There are other things going on in the lab but I’m going to end this here for now… this was supposed to be an introduction after all!
Final comments
*It’s too much work for just the people I currently have and would require too much money for us to study absolutely everything about the LaNts all at once! I’d certainly like to but we have to focus our people and money on certain areas. We are keen to collaborate with anyone who wants to get involved, so get in touch if you are interested!
Self-funded PhD students are welcome to get in touch – we can make a bespoke project involving genetics/genomics, molecular/cellular biology, right up to tissue and whole organism biology depening on what skills you want to develop.
Or if you want to donate to our work directly you can do so via this paypal link, or contact me via the form below.
Some useful links
- Our intro page on laminins
- Our page on LaNts and a cancer switch
- Official lab webpages
- Institute of Ageing and Chronic Disease.
- Some of the lab cartoons #1-5, #6-10
- You might also like our writing guides
About the author
Dr Kevin Hamill is a Senior Lecturer in Cell and Molecular Biology in the Department of Eye and Vision Science, Institute of Ageing and Chronic Disease. at the University of Liverpool. His lab focuses on cell-matrix interactions; you can read about ongoing work elsewhere on this blog, the lab webpages, or published work. As well as supervising PhD, Masters in Research and Undergraduate research projects, Kevin is the academic lead for the Lab skills Module of MRes Clinical Sciences program where he teaches fundamental life scientist skills.
Comments and Questions?
You can also sign up for emails via links in the margins of this page.