If you’ve ever visited the blog before you’ll be aware we do a lot of research on laminins, and you’ve probably seen us talk about basement membranes- part of the extracellular matrix (ECM). If you’re not familiar with the extracellular matrix, trying to picture something as small as a basement membrane or a laminin can be pretty abstract; so I want to quickly take you from ‘human’ through to the basement membrane. If you’re familiar with the ECM, skip straight to the laminin stuff here or the LaNt stuff here.
On the left is a picture of me (or is it Kevin?). I’m a human, and as a human I’m made up of about 30-40 trillion cells, and cells are easy to see under a microscope.
Different types of cells can have totally different shapes, specialised for their specific role in the body. In the photo below, we are looking at cells using a 20X magnification on a Nikon Epifluorescence microscope. These cells are expressing a fluorescent protein called tdTomato that we have made them express so we can study things more easily. In the future I’ll write a blog post about why and how we use fluorescent proteins in our research.
 Inside the cell is where proteins are made, such as laminins, and these can either stay inside the cell or be secreted to the outside. Some of the proteins that cells secrete will make up the “extracellular matrix” (ECM). Examples of ECM structures include cartilage, connective tissue, and of course basement membranes.
Inside the cell is where proteins are made, such as laminins, and these can either stay inside the cell or be secreted to the outside. Some of the proteins that cells secrete will make up the “extracellular matrix” (ECM). Examples of ECM structures include cartilage, connective tissue, and of course basement membranes.
So far, so good.
Basement Membranes
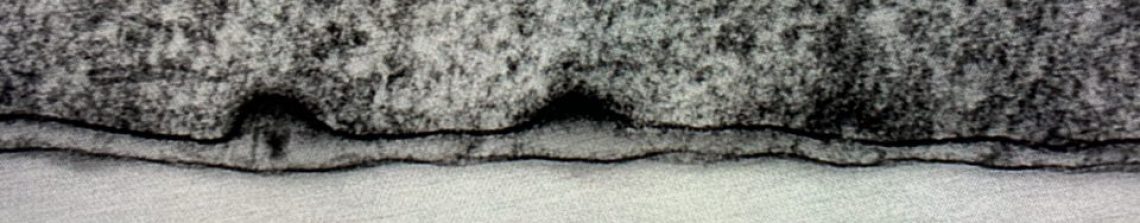
Basement membranes, then. BMs are found in every tissue in the human body. BM proteins evolved approximately 500 million years ago, and BMs were first described in 1840. A long time, right? And we’re still trying to fully understand them! Transmission electron microscopy or immunostaining is usually required to detect basement membranes as their standard thickness is <100nm, with few exceptions – tissues like the lens and trachea need an extra thick BM to function properly.
The small size of most BMs makes it incredibly difficult to model membrane proteins appropriately, and it’s why we are only now beginning to fully appreciate the complexities of BMs. Liam in our lab uses lens basement membranes in his work as they are easier to work with and make a great model system; you can read about how he is learning how to do this here.
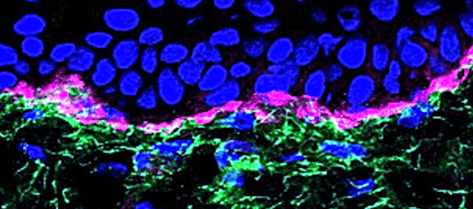
Holding our body together
The main function of BMs has long been thought to be for protecting tissues from breaking apart at the join between cell sheets and the rest of the ECM underneath i.e. protection from mechanical stress. The importance of this function is really clear as when it goes wrong the consequences can be devastating. There are a number of human genetic diseases where one of the BM proteins either doesn’t get made or doesn’t function properly and the effects are really severe. For example, defective BM proteins are associated with a terrible skin blistering disease (epidermolysis bullosa), a muscular dystrophy disorder (MDC1A) or kidney problems (Pierson syndrome and Allport syndrome). Much of what we know about BMs has come from studying these diseases and trying to work toward a treatment for them.
Not just structural support… BMs do a lot more.
In addition to being structural support, BMs are also excellent barriers. They can effectively separate different parts of tissues, for example BMs are part of the blood/brain and blood/retina barriers. They contribute to the control of what passes from one blood supply to the other and have important roles in keeping alive the cells that live on and in even in their structure.
One of the big questions is how do cells and molecules of different sizes pass through this barrier that we call the BM? This process of traversing the BM is particularly important for immune cells to get out of the bloodstream and, no surprise, when cancers metastasize and spread to somewhere else in the body. Understanding more about the BM will eventually let scientists develop better therapies in the future. You can read about one of our projects on this part of BM biology; controlling a cancer-associated switch here.
However, these aren’t the only function of the basement membrane, although this was traditionally the view. There is a growing body of evidence that basement membranes, and the proteins that make up this structure, have much wider roles to play. They can influence cell behaviour in a multitude of ways including telling stem cells to stay as stem cells or directing them to mature into the specific cell types required by different tissues. BMs also have important roles in mechanically sculpting tissues, and providing the road over which cells move during wound repair. Yeah, BMs do a lot!
The video below is one of ours: the black stuff is two proteins we work on being deposited from corneal cells – we’re literally watching them laying their own road. If they do this wrong, the cells won’t move and wounds won’t heal.
What is a Basement Membrane made of?
As BMs have so many different functions it probably won’t surprise you that there is a lot going on down there! Depending on where you go in the body, the components of the BM differ a little bit; not just the thickness but also which proteins are where. However, most are built around two core structural protein families; Type IV collagens and laminins.
Collagen IV and laminins assemble into networks or polymers and it is these networks that provide the key structural support and barrier roles of a BM. Col IV forms a sort of sheet or mat that is pretty robust, whereas laminins assemble into a hexagonal array that is a bit quicker to form but also easier to dissasemble. These two indpendent networks are then cross-linked together through the action of two other core players; nidogens and perlecan. These four groups of proteins aren’t the only things involved, lots of other players are associated with BMs at different times.
I don’t want to go too heavy here so I’ll leave it there for now and write a more in depth discussion of what goes where and when in the future.
Our Research
In the Hamill lab, we are investigating quite a few different aspects of BM and ECM biology. Different group members are asking different questions about how, whey, why what does what in the BM and about what goes wrong in different diseases with the hope that we can identify ways to intervene. For my project, the development of photoswitchable fluorescent proteins, superresolution microscopy techniques and CRISPR-Cas9 gene editing are enabling me to ask new and exciting questions about BM function, assembly and turnover. More detailed info about what I’m doing will be coming in my next post (soon), but for now you can read our introductions to two of our favourite protein families that contribute to BMs multitude of functions; LaNts and Laminins.
Peace and love,
Conor “Conro” Sugden
Conor is a PhD student studying in the Institute of Ageing and Chronic Disease at the University of Liverpool. His work is supported by the Crossley-Barnes bequest studentship and is looking at the role of laminins and LaNts in the lung, in normal and fibrotic conditions.


13 Comments