Explaining the findings of Troughton et al., 2026
A quick recap from Part 1
In our previous studies, we have explored what LaNt proteins are, why the LaNt α31 splice variant is unusual, and how our earlier work showed that it is enriched in the limbal epithelium and upregulated during corneal wound repair. We also saw hints that LaNt α31 influences how epithelial cells spread and migrate.
This new study takes those observations much further — revealing how LaNt α31 changes the behaviour of corneal epithelial cells, and why those changes matter for wound healing and epithelial homeostasis.
What this new study set out to answer
LM332 (formerly laminin‑5) is the dominant laminin in the corneal epithelial basement membrane. It anchors cells via hemidesmosomes and also supports migration during wound repair. But LM332 is unusual: it cannot self‑polymerise like other laminins, so its organisation depends heavily on the cellular environment.
The big question was:
Does LaNt α31 influence LM332 organisation and, through it, the balance between epithelial adhesion and migration?
The answer, as it turns out, is a resounding yes.
Key findings
1. LaNt α31 makes epithelial cells spread more and migrate less
When LaNt α31 was overexpressed in hTCEpi corneal epithelial cells, the cells became more spread — more than double the area of controls — and their migration speed dropped by more than half. This wasn’t due to changes in cell shape or polarity; it was specifically a shift toward a more adhesive, less motile state.
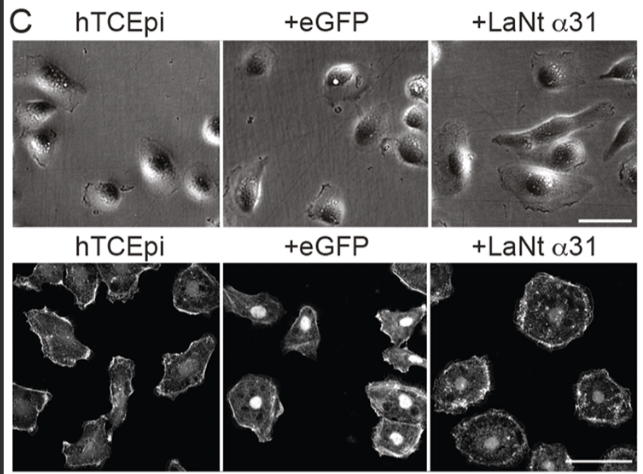
Interestingly, migration could be partially or fully rescued by plating cells on specific extracellular matrices: LM332 or fibronectin → partial rescue, LM511 → full rescue, Collagen I → no rescue. These matrix‑specific effect foreshadows the mechanistic story that follows.
2. LaNt α31 dynamically localises to LM332-rich regions
Using live-cell imaging with LMβ3‑mCherry, the study showed that LaNt α31 forms transient clusters exactly where LM332 is being deposited. These clusters appeared and disappeared rapidly, suggesting LaNt α31 could be involved in organising LM332 rather than simply binding it. Immunoprecipitation confirmed that LaNt α31 and LMβ3 can form a complex (likely indirectly, but we don’t know yet).
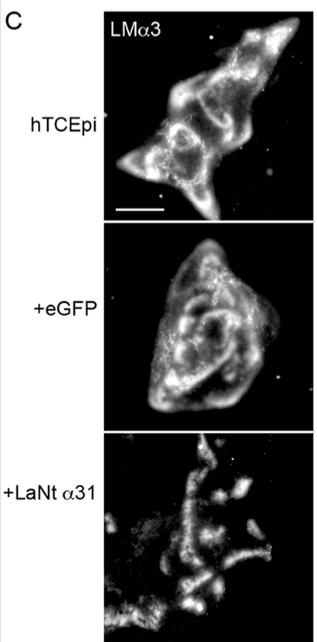
3. LaNt α31 reorganises LM332 into tight clusters
In control cells, LM332 appears as broad arcs and swirls. With LaNt α31 overexpression, LM332 becomes concentrated into discrete clusters. Like stepping stones rather than a path. This suggests a shift in basement membrane architecture. Implying that LaNt α31 acts as a micro‑architectural organiser, shaping how LM332 is presented to the cells that are interacting with it.
4. Hemidesmosomes mature prematurely
The LM332 clustering coincides with accelerated hemidesmosome (HD) assembly. Hemidesmosomes are like spot-welds that control cell adhesion. Our experiments indicate that these adhesions form more rapidly than usual. This is characterised by more colocalisation of β4 integrin with type XVII collagen, increased BPAG1e recruitment at earlier time points, and increased processing of the LMα3 chain (the LG4/5 cleavage associated with HD maturation). Together, these changes indicate that LaNt α31 pushes cells toward a stable adhesion phenotype.
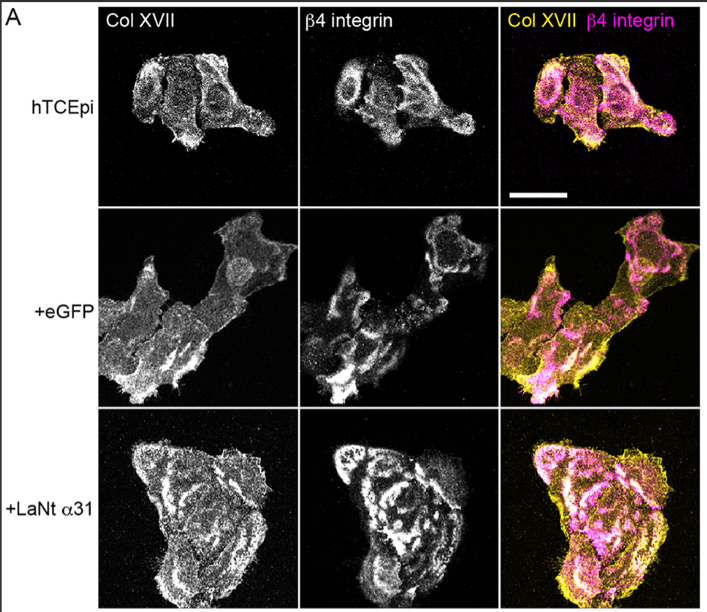
In the pic above you can see the Col XVII in the top two panels is present and in partially co distributes with the b4 integrin in the middle. However, the clustering and co-distribution is increased in the +LaNt a31 cells.
5. Matrix metalloproteinases (MMPs) are essential for these effects
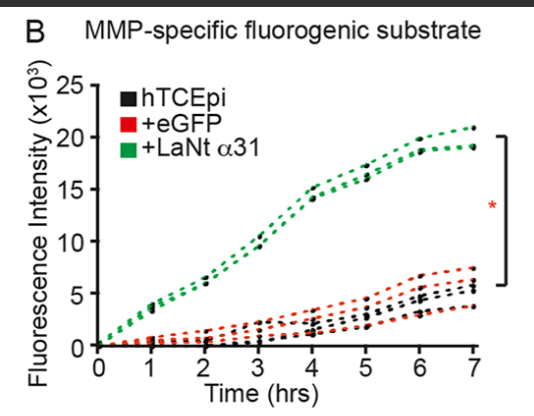
We next asked about the processing event on LM332. We thought it might be driven by matrix metalloproteinases, and testing this led to one of the most striking findings; that increased LaNt α31 cells had increased MMP2/9 activity.
You can see that in the assay below which measured MMP activity, the green lines come from the LaNt cells, the effect was pretty dramatic.
We think that this MMP activity change is the key driver of other aspects of the phenotype, as when MMPs were inhibited, the LM332 clustering disappeared, LMα3 processing returned to baseline, the hemidesmosome maturation was reversed and the cell adhesive phenotype was lost
In other words:
LaNt α31 reshapes the epithelial basement membrane through an MMP‑dependent mechanism.
This positions LaNt α31 as an upstream regulator of proteolytic remodelling — a role not previously appreciated.
What does this mean for corneal biology?
A molecular switch between migration and adhesion
During wound healing, epithelial cells must migrate rapidly, then stop and re‑adhere to restore barrier function. This study suggests LaNt α31 may help control that transition:
- Low LaNt α31 → LM332 remains more diffuse → cells migrate
- High LaNt α31 → LM332 clusters, HDs mature → cells anchor
This aligns beautifully with earlier observations that LaNt α31 is upregulated during wound repair.
Implications for the limbal stem cell niche
The limbus requires a stable adhesive environment to maintain stem/progenitor cells. By promoting HD maturation and LM332 organisation, LaNt α31 may help maintain niche architecture and prevent unwanted migration.
Potential relevance to disease
Because LM332 remodelling and MMP activity are central to many epithelial pathologies, dysregulated LaNt α31 could contribute to:
- recurrent corneal erosions
- delayed wound healing
- fibrosis
- epithelial cancers (consistent with LaNt α31 upregulation in breast cancer)
This opens new avenues for therapeutic exploration.
How this fits into the broader LaNt story
Your earlier work established that LaNt α31 is present, regulated, and biologically active in the cornea. This new study provides the mechanistic backbone:
LaNt α31 is not just a bystander — it is a regulator of basement membrane architecture and epithelial behaviour.
It acts through:
- LM332 reorganisation
- MMP‑dependent LMα3 processing
- Hemidesmosome maturation
- Shifts in the adhesion–migration balance
This positions LaNt α31 as a key modulator of epithelial plasticity.
If LaNt α31 can reshape LM332 architecture this profoundly in vitro, what might it be doing during real corneal injury, repair, or disease? That’s the question we’re chasing next. Keep an eye on this space — the next chapter is already in motion.