Very exciting news – Fight For Sight and DEBRA are supporting a PhD studentship with my group and we are now accepting applications.
From Gene Editing to Therapeutics: Building Human Corneal Models of Inherited Fragility Disorder Junctional Epidermolysis Bullosa
In this project you will work at the frontier of cell engineering and high resolution imaging to understand how inherited mutations cause the cornea to break down in junctional epidermolysis bullosa. It offers an exciting mix of discovery biology + translational model development, with clear onward routes toward future therapies.
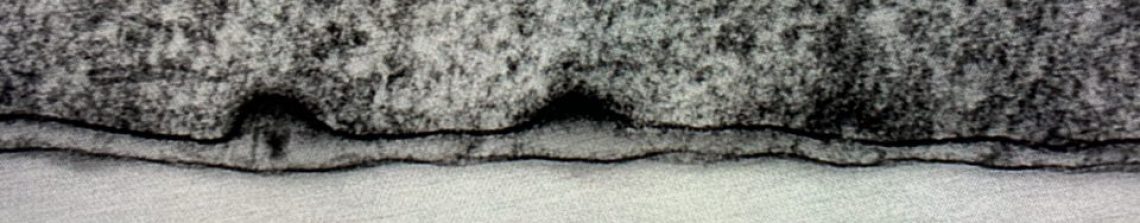
Junctional epidermolysis bullosa (JEB) is one of the most severe inherited blistering disorders. From birth, patients have incredibly fragile skin and other epithelium, with associated painful blisters, chronic wounds. It is brutal. For many patients the eye is affected early and relentlessly. Even small amounts of friction can cause the corneal surface to break down, leading to painful erosions, repeated wounds and progressive scarring that can ultimately result in blindness.
The reason these problems arise is that patients have mutations that affect the function or expression of the proteins involved in epithelial cell-to-extracellular matrix adhesion (hemidesmosomes). In JEB, the protein most usually affected in laminin 332 which has long been established as essential for anchoring the corneal epithelium. It is a really interesting protein that my lab has focused on for many years (you can read our simple introduction to laminins here). In this project we aim to harness improvements in technology to generate high‑resolution understanding of how patient‑specific mutations disrupt laminin 332 assembly and function, and, at the same time develop laboratory platforms for testing emerging treatments using a combination of advanced imaging, gene editing and human 3D corneal tissue models.
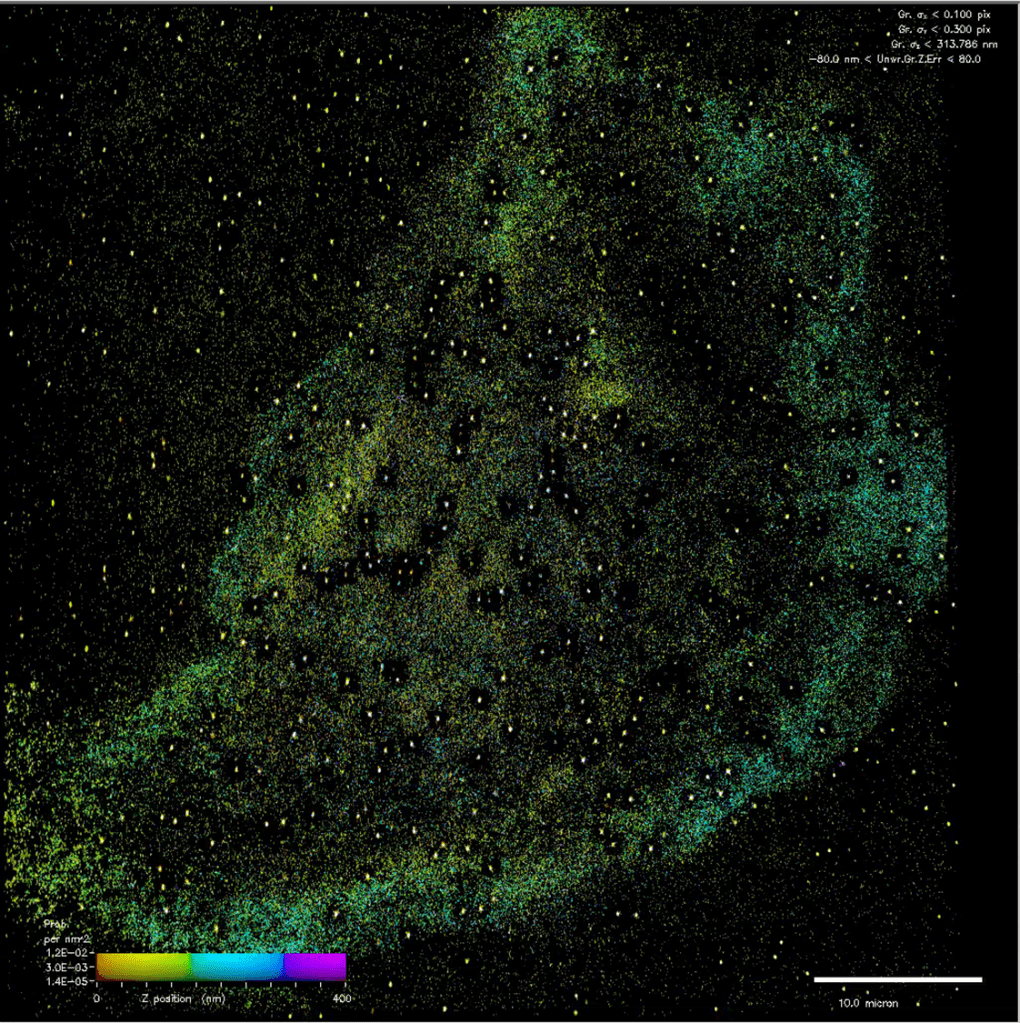
Laminin‑332 is built from three subunits, and mutations in the LAMB3 gene are the most common cause of JEB. Recent advances from our research team have now make it possible to fluorescently tag endogenous laminin‑332 in human corneal cells without disturbing its function. At the bottom of the page you can watch a webinar I recently delivered on some of this work.
Building on our new cool tools, this project will use CRISPR/Cas9 genome editing to engineer precise versions of JEB mutations, in combination with super‑resolution imaging methods and live‑cell photoconversion microscopy. These techniques will allow us to visualise individual laminin molecules, map their organisation in the basement membrane and watch how cells attempt to repair epithelial damage in real‑time. The goal is to uncover the nanoscale mechanisms of corneal fragility and identify exactly how different mutations compromise tissue strength and wound healing.
A major component of the project involves creating next‑generation human models of JEB. The student will develop 2D cultures and then advanced 3D corneal-equivalents using engineered collagen scaffolds and biofabricated substrates optimised for imaging. These models will enable the student to compare healthy versus mutant cells in environments that closely mimic the human eye.
Our new models will form the foundation of a powerful therapeutic screening system: if a drug or gene‑based therapy successfully restores laminin‑332, the cells will produce a quantifiable read-out of rescue. The aim is to create an accessible, scalable platform for testing candidate treatments, helping to accelerate the search for effective therapies for patients.
Throughout the PhD, the student will receive training in CRISPR genome editing, high‑resolution microscopy, 3D tissue engineering, advanced image analysis, and quantitative screening. They will work within a multidisciplinary team combining cell biology (Dr Hannah Levis), matrix biochemistry (Dr Kevin Hamill), biomaterials engineering (Dr Lucy Bosworth) and clinical expertise in corneal disease (Prof Stephen Kaye). The project is designed to give flexibility and ownership, allowing the student to focus on mechanisms of basement membrane assembly, model development, wound‑healing biology, or translational assay design depending on their interests.
By the end of the project, the student will have created a unique, human‑relevant disease model for JEB, generated new biological insight into laminin‑332 organisation, and delivered a screening platform that can be used to evaluate emerging drug and gene therapies. This is a rare opportunity to work at the interface of cutting‑edge imaging and meaningful patient‑centred translational research, ideal for students excited by discovery science with a clear pathway to improving future treatments for sight‑threatening disease.
Get in touch! Send me an email – khamill@liverpool.ac.uk to set up a time to chat about the project.
This is a funded project that will pay tuition fees at the UK home rate, provide a stiped at the current UKRI rate and provides some support for experimental costs.