I’m very pleased to announce that our recent work has been published in the journal Genes (click the image below to reach the article, open access)
This work really represents the major thrust of John Knox‘s PhD studies. Indeed, the four authors are John and his supervisory team of myself, Prof George Bou Gharios (University of Liverpool) and Prof Colin Willoughby (University of Ulster). So, really, this is a big shout out for John for seeing all his work come to fruition.
While the title is a bit of a mouthful, the underlying “story” is quite simple.
Glaucoma is one of the leading causes of blindness worldwide. One of the main drivers of the disease is blockage of the drainage channels in the eye which leads to increased internal pressure. This pressure is what gets measured at the opticians and too much pressure ultimately causes degeneration of the optic nerve and progressive loss of vision.
The cells that line the drainage channel are known as Trabecular Meshwork (TM) cells. They undergo a bunch of changes on their route to dysfunction. However, the best characterised and biggest driver of these pathogenic changes is the protein transforming growth factor beta2 (TGFB2). TGFB2 acts via many routes but in the TM its main partner proteins (effector protein) is known as connective tissue growth factor (CTGF).
Here we hypothesised that we could help protect TM cells against excessive TGFB2 by selectively targeting its effector protein CTGF. We’re not the first to suggest that; however, our approach was a little different. We wanted to use a “natural” way to turn down CTGF.
One of the way genes are controlled is through the action of micro RNAs (miRs). These are exactly what they sound like; small RNAs that bind onto specific target regions on messenger RNAs and block their translation.
John used a series on tools to hunt for miRs that could target CTGF, then he analysed how stable those miRs are across multiple human donor derived TM cells. From those experiments miR-18a fell out as the most consistent in terms of response to TGFB2.
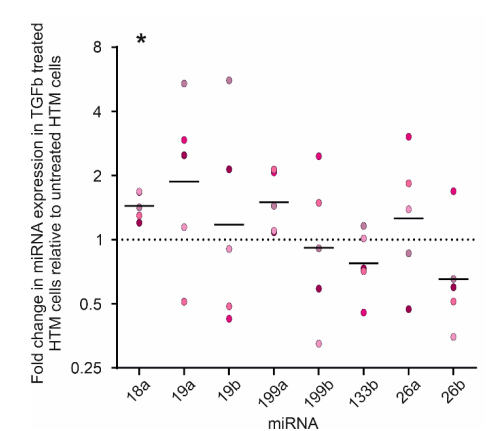
Based on these data, John developed the use of a mimic of miR-18a as a potential treatment for the TM cells. In these experiments, for comparison, he used a custom designed (i.e. not naturally occurring) miR equivalent (a shRNA) to target CTGF. Treatment of TM cells with both the natural and synthetic treatment protected the cells against TGFB2-induced upregulation of CTGF protein and, importantly, against TGFB2-induced contraction. In other words, the miR 18a mimic treatment looks like a good candidate to develop into a therapy.
One of the cool things about this miR approach is that most miRs have evolved to hit multiple targets within a linked pathway, and one reason why see slightly better outcomes with miR18a treatment is likely due to this phenomenon, we’re not just hitting CTGF but also other downstream effectors.
miRNA therapies are likely the future for a lot of disease and identifying which ones to use and when is an essential step toward developing a viable strategy. From here we need to overcome are how to delivery to the right cells at the right time and determine whether we can develop a combination therapy to increase potency.
This work was funded by Fight for Sight, an eye charity that supports research into all eye and vision disorders.



1 Comment