Read the published paper here or read on for an overview.
Synopsis
- Many diseases destroy the conjunctiva, the white part of the eye, meaning that we need to develop ways to replace the damaged tissue. So, we wanted to find out the best way to grow conjunctival cells.
- We did a series of experiments that showed that conjunctival cells deposit proteins that help their growth, and that the best mix of proteins comes from cells in culture for short periods of time.
- We catalogued all the proteins deposited by cells at different time points, providing a resource that researchers can use in the future.
- The data revealed differences in the proteins present at different time points, including a protein that had not been investigated in the conjunctiva before, a2-hs-gp. We tested a2-hs-gp on its own, and it turned out to be a good substrate for conjunctival growth.
- These findings will help to accelerate the development of new therapies.

The full story…
A real problem.
The white part of the eye, the conjuntiva, is covered by layers of epithelial cells that are really important in keeping the eye healthy. If you’ve ever had conjunctivitis (pink eye) you already know this; the epithelia on the conjunctiva is a crucial barrier to protecting the eye from bacterial and viral infections.
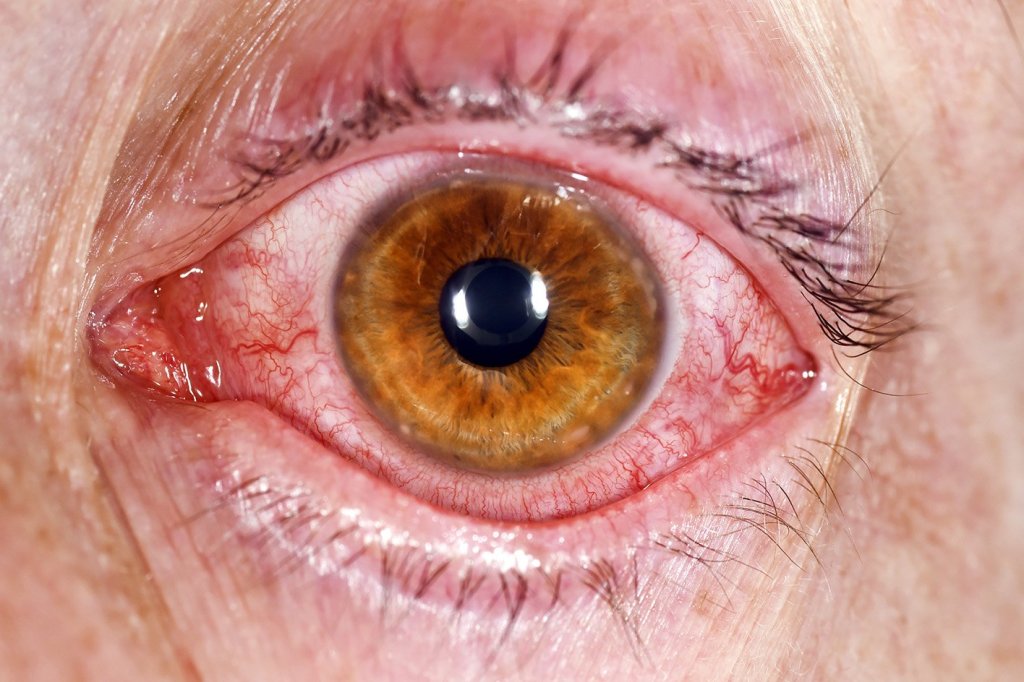
Conjunctivitis is itchy and painful but usually treatable with drops; however, there are lots of other situations where defective conjunctiva require surgical intervention for example after chemical burns. Actually, it’s crucial that the conjunctiva is healthy before other procedures on the front of the eye are carried out as things like corneal grafts will fail if the conjunctiva is not repaired first.
But there’s a problem. There aren’t enough healthy conjunctivas being donated to meet the clinical need. One solution is to encourage more people to donate (let your family know that you want to improve the lives of others by helping them to see after your death). An additional solution is to develop ways in which we can make the donated material we have available stretch further by developing techniques to expand the cells from one donor so that they can be used to treat 2, 3, 4, + patients.
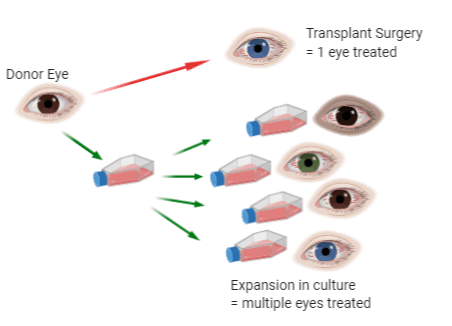
Sounds simple enough, but conjunctival cells aren’t very easy to grow! So, we set out to discover if we could identify any ways to improve the knowledge of what makes a conjunctival epithelial cell happy and thereby identify ways to make the cells easier to grow.
Ask the cells?
The problem of conjunctival shortage and the difficulties in culturing cells have been known for a long time, and so many things have been tried already by some really excellent researchers around the world. Our starting point, therefore, was made easier as we could stand on the shoulders of those that came before us. However, science progresses and techniques are available now that meant we could ask our question in a different way.
Our idea was quite simple. The best source of information about what works for growing conjunctival cells is… conjunctival cells! So, we asked them.
When cells grow they modify their environment by depositing a selection of proteins onto the surface. We describe these outside-of-the-cell proteins as the “extracellular matrix”. Once deposited, the extracellular matrix provides the points for cells to attach and the extracellular proteins controls a whole host of cellular activities including growth, attachment, movement, differentiation, regulation of cell shape and polarity. Indeed, the extracellular matrix is absolutely key in defining what cells become, for example a difference in extracellular matrix can help determine whether an epithelial cell becomes skin, cornea or, in this case, conjunctiva.
Full disclaimer, I consider myself to be a matrix biologist, and as such I always think the answer to most questions lies in the proteins deposited by cells!

Pop leaves the matrix
So we set about finding out what extracellular matrix conjunctival cells make and what they like. To do this, Aruni Makuloluwa, as part of her PhD studies, cultured a well-characterised conjunctival cell line (developed by our collaborators at Harvard, Ilene Gipson who is one of the pioneers in conjunctival research). Aruni grew these cells on a polystyrene dish for different periods of time, allowing them the opportunity to lay down an extracellular matrix and continue to modify that matrix over time.
She then burst the cells open by hyperosmotic shock and washed away the cellular material to leave behind just the matrix proteins. Then she used that isolated matrix for two different sets of experiments,
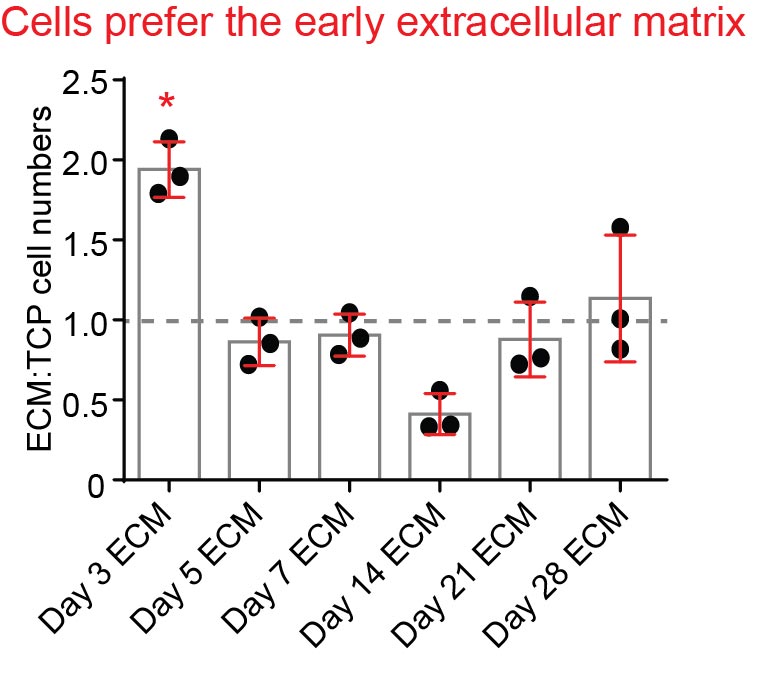
First, Aruni asked whether the cell-derived extracellular matrix was better at supporting conjunctival cell attachment and growth than tissue culture plastic (it was, as we had hoped and expected. Phew!). She also asked whether there was any difference between the matrixes from short-term vs long-term culture, and it turned out there was; the cells showed a clear preference for the matrix derived from cells that had been on the dish for 3 days compared with the longer time points.

Next, she asked what proteins were present in each matrix using mass spectrometry. These data are really cool. They tell you just about everything that is present and their relative abundance, and because we analysed multiple time points, it also could tell us how the matrix changed. The data represent a useful resource for others working in this area in the future (downloadable via the journal site).
Analysis of the mass spec data revealed that (as expected) the most abundant matrix proteins were, my favourite, the laminins and of that family mostly laminins alpha3 , beta3, and gamma2 (we also pulled out my favourite laminin-binding proteins, integrins alpha6 and beta4). Combined, these accounted for almost half of the proteins detected at all time points. This was a good sign, when you get what you expect, what is supported by previous work in this area, then you can be pretty confident that the techniques are working! So far so good. But the real question was what proteins were different between the different time points.
a2-HS-GP
When we analysed the data looking specifically for proteins that were highly expressed in the early timepoints compared with the late and one protein (you guessed it) the alpha2-Heparan Sulfate-glycoproptein (a2-HS-GP aka fetuin-A) jumped out as a pretty striking candidate.
At our earliest time point a2-HS-GP consisted on ~5% of the peptides detected, and when combined with binding partners that rose to about 17%. However, the kicker was, that from day 7 onward the levels dropped to almost non detectable levels.
I’d never heard of a2-HS-GP and it had never been associated with culturing epithelial cells. This really highlights the power of the unbiased approach we took here. If we had restricted our studies to just the things we knew about, then we would never have tested a2-HS-GP. Or, to be completely accurate, we would never even have considered it as an option. [There are other proteins which changed in similar or opposing ways, those details might be important too and are described in the paper].

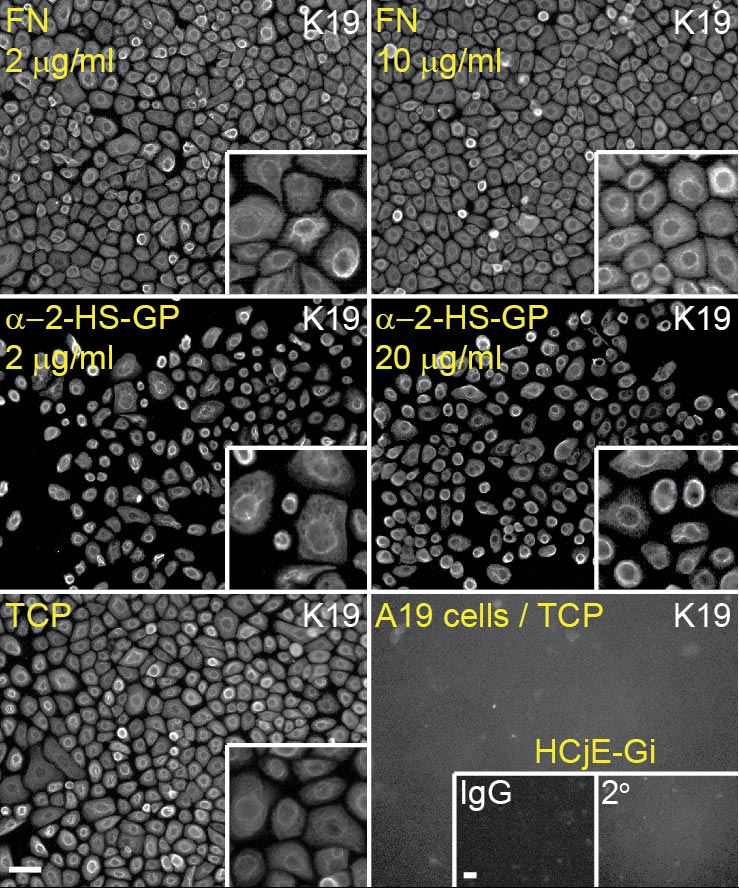
As every science teacher will tell you; correlation does not necessarily imply causation. So next, Aruni coated dishes with different concentrations of just a2-HS-GP and tested to see how cells responded to it (in the images below fibronectin (FN) is used as for comparison.
The data revealed that a2-HS-GP isn’t really sticky; the cells attached just as well to a2-HS-GP coated dishes as they did to uncoated cell culture dishes (unlike FN which is like glue!). However, on the a2-HS-GP the growth rates increased in a concentration-dependent manner. Pretty much exactly what we were looking for. 🙂
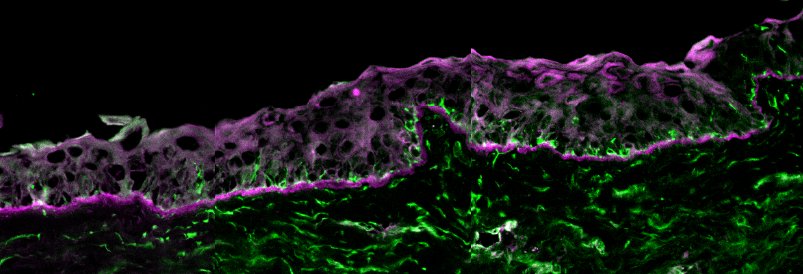
We also checked if the cells stayed conjunctival-like or, which would be a major problem, they converted into other cell types. The images below come from one such check, where we have examined the expression of protein that is expressed highly in conjunctival cells. The finding that the cells still express this protein when plated on a2-HS-GP is a good sign that the cells have retained their conjunctival features.
How will we use this information?
To summarise; these studies have identified a “new”* protein that can be used to help the growth of conjunctival cells, and has provided a useful resource; the catalogue of all the proteins deposited by conj cells in culture.
Knowing that this protein can help the growth of conjunctival cells means that we can now start to exploit that information in our ongoing work.
At the University of Liverpool, we have a number of research groups working on making artificial substrates for transplantation (including Prof Rachel Williams, who was instrumental in directing this research, as well as Dr Hannah Levis, Dr Atikah Haneef, Dr Victoria Kearns, Dr Lucy Bosworth and others). Its one of the areas where the Department of Eye and Vision Science really excels. Adding a2-HS-GP to the new or improved substrates that are being developed locally or world-wide could (should!) help conjunctival growth. Which would mean that we are one step closer to the clinic.
Once we step forward, our clinical colleagues are in place to push things even further. Part of the research team here included Prof Stephen Kaye, a consultant ophthalmologist and Professor of clinical ophtalmology at St Paul’s Eye Unit in the Royal Liverpool Hospital, and Dr Rosalind Stewart a corneal consultant at Leeds Hospital. Bringing together the bench science with the tissue engineers and clinical academics means our research journey from bench to bedside is mapped out.
The road to discovery is a journey. Realistically, this paper is somewhere in the middle of that journey. But every step is important and we think that the findings described here are a valuable advance.
Support our work
If you would like to help drive forward research into new treatments for eye conditions, consider donating to St Paul’s Research Foundation .
Read the paper here
*the protein itself isn’t new, this discovery is a protein that hasn’t been investigated in the conjunctiva before.


1 Comment